Positive Health Online
Your Country

Letters to the Editor Issue 274
listed in letters to the editor, originally published in issue 274 - November 2021
Dear Scientific and Medical Network Readers
The Scientific and Medical Network carries reviews of over 200 books a year in Paradigm Explorer, mostly by myself as editor. A database maintained in Scotland between 1996 and 2014 contained over 5,000 entries! At present, there is no way of searching for past book reviews, so we are in the process of creating a searchable database of historic reviews as a form of outreach that will be available as a new member benefit.
We have identified a suitable Wordpress plug-in for the data, and Kevin Ashbridge is working on uploading an initial batch going back continuously to 2013, plus 2002-2004 and an additional twelve issues. The reason for these gaps is that the hard drives onto which the issues had been uploaded ceased to be accessible. In the second phase we will work on extracting the rest of the reviews from pdf files going back from 2011. We will make reviews from one back issue publicly available as a sample.
As potential beneficiaries of this service we would hugely appreciate your financial participation and support. Our target is £5,000 and we have an initial contribution of £735 from a restricted fund to put towards this.
Please consider supporting this by clicking on the button below.
Any donation, whether big or small, makes a difference to our mission.
https://www.paypal.com/donate/?cmd=_s-xclick&hosted_button_id=Y2SJX7EL277ME&source=url
Many thanks in anticipation for your support.
With every good wish,
David Lorimer
Programme Director, Scientific and Medical Network
Editor, Paradigm Explorer
Southampton Scientists Discover a Fundamental Feature of Aggressive Lymphomas
Research led by the University of Southampton has revealed a new fundamental feature of aggressive B-cell lymphomas which could open the door to further research into early detection and treatment of the disease.
Over 14,000 people are diagnosed with lymphoma each year in the UK, making it the fifth most common cancer and the most common blood cancer (Source: Blood Cancer UK).
In this new study, a team led by Prof. Francesco Forconi, identified a tumour-specific change, not seen in normal B cells. B cells are part of the human body's immune system and are responsible for producing antibodies; they display an antibody-like molecule on their surface, known as the B-cell receptor. The new findings have shown how the receptors can differ in aggressive lymphomas by the presence of unusual sugars, known as glycans, in the antigen-binding sites of the lymphoma B-cell receptor.
The findings have been published in Blood, the Journal of the American Society of Haematology.
The team, which included glycobiologist Prof. Max Crispin and cancer immunologist Prof. Freda Stevenson, have revealed that these glycans have a specific structure that allow the lymphoma cells to receive signals from molecules called “lectins”, which are attached to surrounding cells, enabling the tumour to survive - and grow - in the lymph nodes.
Francesco Forconi, Professor of Haematology at the University of Southampton said, “this very exciting team-work describes the structure of the glycans covering the surface of the tumour's B-cell receptor and how it works. It is a remarkable tumour-specific feature required by all the tumour cells of patients with the most common lymphomas.”
Prof. Forconi continued, "this is a new specificity required by the lymphoma cells to survive which we now know how to detect and are learning how it functions. Our findings are paving the way to further investigations, including early cancer detection and therapeutic targeting, both of which will be our future goals".
The study has been funded by Blood Cancer UK charity and the Keanu Eyles Fellowship. The next steps will be to precisely target the interactions between these glycans and the lectins by therapeutic antibodies that are being developed by Forconi's team in collaboration with the Antibody Vaccine Group at The University of Southampton and Professor Carl Figdor at Radboud University in the Netherlands in a Cancer Research UK funded project.
The joint work between Professor Forconi's lab, which specialises in analysing B-cell receptors in leukaemias and lymphomas, and Professor Crispin's lab, which has expertise in the structure of glycans, will continue, bringing scientific and clinical specialities closer together.
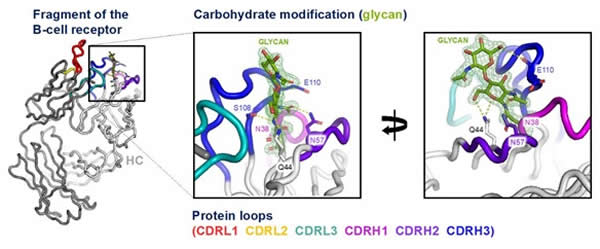
The image depicts the crystal structure of a fragment of a B-cell receptor from an aggressive lymphoma. The receptor is modified by carbohydrate, termed a glycan, shown in green sticks. The protein chains are depicted as a tube with the loops around the glycan coloured.
Further Information
For further information and interview requests, please contact Steve Bates, Media Relations Officer, University of Southampton, s.d.bates@soton.ac.uk ,Tel: 07342 060429.
Reference
Insertion of atypical glycans into the tumor antigen-binding site identifies DLBCLs with distinct origin and behavior has been published in Blood with DOI https://doi.org/10.1182/blood.2021012052
Dementia Risk in Former Professional Footballers Related to Player Position and Career Length
The landmark, University of Glasgow-led research into lifelong health outcomes in former professional footballers, the FIELD study, has revealed further major insights into the link between football and risk of dementia. In findings published in the journal JAMA Neurology,[1] researchers report that risk of neurodegenerative disease in former professional footballers varies by player position and career length, but not by playing era. These results add to the ground-breaking observations from the 2019 FIELD study publication, which found that former professional footballers had an approximately three-and-a-half-fold higher rate of death due to neurodegenerative disease than expected.
Led by consultant neuropathologist Professor Willie Stewart, Honorary Professor at the University of Glasgow, the researchers looked at health records data for around 8,000 Scottish former professional footballers and 23,000 matched general population controls and explored whether risk of neurodegenerative disease varied by player position, length of career or playing era. The results show that, for goalkeepers, neurodegenerative disease risk was similar to general population levels. In contrast, neurodegenerative disease risk for outfield players was almost 4 times higher than expected and varied by player position with risk highest among defenders, at around 5-fold higher than expected.
These new findings also show that neurodegenerative disease diagnoses increased with increasing career length, ranging from an approximately doubling of risk in those with shortest careers, to around a 5-fold increase in those with the longest careers. However, despite changes in football technology and head injury management over the decades, there is no evidence neurodegenerative disease risk changed for the population of footballers included in this study, whose careers spanned from around 1930 to the late 1990s.
Prof Willie Stewart said:
“We have already established that former professional footballers are at a much greater risk of death from dementia and other neurodegenerative disorders than expected. These latest data from FIELD take that observation further and suggest this risk reflects cumulative exposure to factors associated with outfield positions.”
In parallel work led by Prof Stewart, a specific pathology linked to brain injury exposure, known as chronic traumatic encephalopathy (CTE), has been described in a high proportion of the brains of former contact sport athletes, including former amateur and professional footballers.
Prof Stewart added:
“Taking these new results together with our and others post-mortem findings and data from our previous FIELD studies, the evidence is clear that the standout risk factor for neurodegenerative disease in football is exposure to head injury and head impacts. As such, a precautionary principle approach should be adopted to reduce, if not eliminate exposure to unnecessary head impacts and better manage head injuries in football and other sports.”
Charlotte Cowie, FA Chief Medical Officer, said:
“The FIELD study team, funded by ourselves and the PFA, have continued to produce insightful data that has enabled us to make changes in the game. We welcome these new findings.”
PFA Chief Executive, Maheta Molango, said:
“The PFA would like to thank the FIELD study team. The welfare of our players, past, present and future is at the forefront of everything we do and this data will inform us how best to protect them and improve our services.”
FIELD is the largest study to date looking in this detail at the risk of neurodegenerative disease in any sport, not just professional footballer players. The study compared health records of 7,676 former Scottish male professional football players who were born between 1900 and 1976 against those of more than 23,000 matched individuals from the general population. Neurodegenerative disease risk among former football players relative to matched controls was then calculated for a range of player positions and career lengths and for decade of birth.
The work is supported by funding from The Football Association and The Professional Footballers’ Association Charity; U.S. National Institutes of Health, National Institute of Neurologic Disorders and Stroke; and NHS Research Scotland.
Further Information on the Study
- 2/3rd of the footballers included in this study were born after 1952;
- The study used electronic health records to compare data on hospitalisation, prescribing for dementia and causes of death in Scottish former professional football players;
- Study inclusion was restricted to individuals born prior to 1st January 1977, with study follow-up commencing from age 40. All former football players were male;
- Average career lengths for outfield players included in FIELD were around 8.6 years, with professional career typically commencing around age 20. As such, players born in 1969 would typically have careers spanning 1989 to 1998;
- Over the follow-up period, 5% (386/7676) of former football/ soccer players were identified with neurodegenerative disease, compared to 1.6% (366/23,028) of matched population controls;
- The FIELD study is an acronym for ‘Football’s InfluencE on Lifelong health and Dementia risk.’;
- This is the sixth output from the FIELD study, the previous five being:
- The aforementioned mentioned landmark FIELD study on former professional footballers neurodegenerative disease risk, published in the New England Journal of Medicine https://www.nejm.org/doi/full/10.1056/NEJMoa1908483
- The FIELD results showing that former professional football players have lower risk of hospitalization for the most common mental health disorders, including anxiety and depression, and are at no greater risk of suicide: https://jnnp.bmj.com/content/91/12/1256
- A manuscript detailing brain pathology in former footballers and rugby players with dementia in Acta Neuropathologica https://doi.org/10.1007/s00401-019-02030-y
- Research looking at specifics of CTE pathology in Brain Communications https://academic.oup.com/braincomms/advance-article/doi/10.1093/braincomms/fcaa210/6017821
- The FIELD study protocol, published in BMJOpen http://dx.doi.org/10.1136/bmjopen-2018-028654
Reference
Russell ER, Mackay DF, Stewart K, MacLean JA, Pell JP, Stewart W. Association of Field Position and Career Length With Risk of Neurodegenerative Disease in Male Former Professional Soccer Players. JAMA Neurol. Published online August 02, 2021. doi:10.1001/jamaneurol.2021.2403. https://jamanetwork.com/journals/jamaneurology/fullarticle/2782750
Further Information
For more information contact Elizabeth McMeekin or Richard Warburton in the University of Glasgow Communications and Public Affairs Office on Tel 0141 330 4831; Elizabeth.mcmeekin@glasgow.ac.uk or richard.warburton@glasgow.ac.uk
Viral Mutations and the Risk of 'Second-hand Malnutrition'
by Michael Passwater
Republished from orthomolecular.activehosted.com
The New York Times recently quoted Michael Osterholm, an expert epidemiologist at the University of Minnesota, acknowledging, "We still are really in the cave ages in terms of understanding how viruses emerge, how they spread, how they start and stop, why they do what they do."[1] While it is true of many topics that new studies often lead to more questions than answers, this topic has special importance to the human experience. We must accelerate our understanding of the complex interactions between humans and viruses to survive and to improve our experience on Earth.
Fortunately, over the past three decades, brilliant pioneers including Ethan Will Taylor, Melinda Beck, and Caroline Broome have been diligently pointing the way out of the cave. The biochemistry is complicated, the genetics is even more complex, and the terminology is unfamiliar. But the message is too important to hide in a library. It demands attempts to understand and communicate their findings. It may require a shift in thinking, and a shift in actions.
Readers of the Orthomolecular Medicine News Service appreciate the importance of good nutrition to keep our bodies healthy. Let's shift our attention for a moment to explore the impact of nutrition on an invading virus.[2,3] RNA viruses are responsible for many of the most devastating infectious diseases of our time – Ebola, Dengue, Influenza, Hepatitis C, Polio, Zika, SARS, MERS, and SARS-CoV-2, among others. However, in addition to being so incomplete that they are fully dependent on invading host cells to replicate themselves, RNA viruses are also primitive in ways that make them unstable. Their replication process is rapid, error-prone, and devoid of the nucleic acid and protein folding proof-reading enzymes used by advanced organisms to minimize mutations and mis-translations during nucleic acid replication and protein production. As a result of this instability, truly pure strains of a virus are rarely found in nature. Viruses tend to exist as mixtures of closely related variants, sometimes referred to as a "quasi-species".
Melinda Beck and colleagues performed experiments with coxsackie and influenza viruses exploring the impact of host nutrition on these viruses. [4-11] They injected a virus (quasi-species) considered to be avirulent (not producing illness) into nourished mice (specifically, selenium and vitamin E sufficient mice), and injected the same virus into selenium and vitamin E deficient mice. The nourished mice did not become ill, and consensus viral genomes from these mice reflected the initial avirulent virus injected. Accordingly, injecting the virus harvested from the first round of nourished mice into additional nourished mice continued to not produce illness. However, the malnourished mice injected with the initial virus died, and virus isolated from these mice were found to have mutations increasing their pathogenicity. This mutated virus was then injected into nourished mice, and they died too.[6,7]
Caroline Broome and colleagues explored this concept in humans using oral live attenuated poliomyelitis vaccines.[11] The test groups were given 0, 50 or 100 micrograms (mcg) of selenium as sodium selenite daily for 6 weeks before being exposed to the attenuated virus, and for 9 weeks following exposure. The people receiving 100 mcg of selenium per day cleared the virus sooner, and virus isolated from this group contained fewer mutations.
The researchers suggested three factors as possible explanations of their findings:
- Decreased immune function in the selenium and vitamin E deficient mice, allowing minor populations of more virulent strains of the overall avirulent quasi-species to escape eradication by the immune system.
- A shift in the intracellular redox balance towards oxidation, allowing faster viral replication. Lower intracellular glutathione concentrations are associated with higher viral titres.[12,13]
- Increased oxidative stress leading to new viral mutations as a result of direct oxidative damage to the viral RNA.
Selenoproteins are important for immune function, including interferon production, phagocytosis (white blood cell destruction of invading virus or other pathogen), and the creation and maintenance of immune memory cells. Without adequate selenoproteins, immune responses to infections and vaccinations are suboptimal and resulting cellular and humoral immunity, if achieved, lasts for a shorter duration.[11,14] Ethan Will Taylor and colleagues have shown that RNA viruses destroy host selenoproteins and other components of DNA synthesis to favour self-replication of the RNA virus.[15-18] Like pirates taking a seaside village's treasures and using it for their own purposes, an RNA virus can disrupt the host cell's defences and use the host nutrients, nucleic acids, and assembly mechanisms to mass produce itself. In 2009, Dr Taylor detailed the cellular metabolism disruption that results from HIV and other RNA viruses leading to NAD+ depletion, ATP depletion, and necrosis.[15] In some cases, immunosuppression also results due to depletion of the tryptophan pathway.
Glutathione peroxidases and thioredoxin reductases are selenoproteins essential as direct antioxidants, and to recycle other antioxidants such as ascorbic acid (vitamin C) and tocopherols (vitamin E).[19] Ascorbic acid, in turn, also helps to minimize nucleic acid mutations, and to maintain redox balance among its many roles throughout the body. Reactive oxygen species (O2-) and reactive nitrogen species (NO, ONOO-) play a large role in causing RNA virus mutations, and also damaging host cells and nucleic acids.[12,13] Pre-infection insufficiency or disruption (acquired deficiency induced by the virus) of the host antioxidant network increases the severity of the current illness, and leads to RNA virus mutations which increases the risk of severe illness to other hosts in the future.[20-28]
Maybe more is known about the emergence of pathogenic viruses than we realize? It turns out the same good nutrition that keeps people strong, also keeps viral genomes more stable and less pathogenic. [29-35] Similar to the unpleasant reality that a non-smoker frequently in proximity to a smoker may become ill from second-hand smoke, a well-nourished person may be vulnerable to illness from exposure to mutated viruses arising from viral replication in malnourished hosts.
Addressing micronutrient as well as macronutrient insufficiencies and imbalances throughout our communities and food chains is critical to public health.
Recommended adult doses to reduce risk of serious viral infections[28-34]:
- Vitamin C, 500-1000 mg, 3 times daily (more to bowel tolerance if sick)
- Vitamin D3, 5,000 IU (125 mcg)/day (maintain plasma vitamin D level in the 40 - 80 ng/mL range)
- Vitamin K2, 100 mcg/day
- Niacin / niacinamide 200 - 1000 mg/day (in divided doses, start with smaller doses, increase over weeks)
- Magnesium 400 mg/day (in malate, citrate, chelate, or chloride form)
- Zinc, 20 mg/day
- Copper 2 mg/day (along with zinc, in chelate, orotate, or gluconate form)
- Selenium 100-200 mcg/day
- Vitamin E 400 IU (268 mg)/day
References and Further Reading
- Leonhardt D, Wu A (2021) "Has Delta Peaked?" The New York Times, The Morning. September 1, 2021. https://www.nytimes.com/2021/09/01/briefing/delta-peak-covid-caseload.html
- Beck MA, Handy J, Levander OA (2004) Host nutritional status: the neglected virulence factor. Trends Microbiol 12:417-423. https://pubmed.ncbi.nlm.nih.gov/15337163
- Guillin OM, Vindry C, Ohlmann T, Chavatte L (2019) Selenium, Selenoproteins and Viral Infection. Nutrients 11:2101. https://pubmed.ncbi.nlm.nih.gov/31487871
- Beck MA (1999) Trace Minerals, Immune Function, and Viral Evolution. Chapter 16 in: Military Strategies for Sustainment of Nutrition and Immune Function in the Field. Institute of Medicine (US), Committee on Military Nutrition Research. US National Academies Press. ISBN-13: 978-0309063456 https://www.ncbi.nlm.nih.gov/books/NBK230971
- Beck MA. Kolbeck PC, Rohr LH, et al. (1994a) Benign human enterovirus becomes virulent in selenium-deficient mice. J. Med. Virol. 43:166-170. https://pubmed.ncbi.nlm.nih.gov/8083665
- Beck MA, Kolbeck PC, Rohr LH, et al. (1994b) Vitamin E deficiency intensifies the myocardial injury of coxsackievirus B3 infection of mice. J. Nutr. 124:345-358. https://pubmed.ncbi.nlm.nih.gov/8120653
- Beck MA, Shi Q, Morris VC, Levander OA. (1995) Rapid genomic evolution of a non-virulent coxsackievirus B3 in selenium-deficient mice results in selection of identical virulent isolates. Nat. Med. 1:433-436. https://pubmed.ncbi.nlm.nih.gov/7585090
- Nelson HK, Shi Q, Van Dael P. et al. (2001) Host nutritional selenium status as a driving force for influenza virus mutations. FASEB J. 15:1721-1738. https://pubmed.ncbi.nlm.nih.gov/11481250
- Beck MA, Nelson HK, Shi Q, et al. (2001) Selenium deficiency increases the pathology of an influenza virus infection. FASEB J. 15:1481-1483. https://pubmed.ncbi.nlm.nih.gov/11387264
- Beck MA, Levander OA. (1998) Dietary oxidative stress and the potentiation of viral infection. Annu. Rev. Nutr. 18:93-116. https://pubmed.ncbi.nlm.nih.gov/9706220
- Broome CS, McArdle F, Kyle JAM, et al. (2004) An increase in selenium intake improves immune function and poliovirus handling in adults with marginal selenium status. Am. J. Clin. Nutr. 80:154-162. https://pubmed.ncbi.nlm.nih.gov/15213043
- Akaike T, Fujii S, Kato A, et al. (2000) Viral mutation accelerated by nitric oxide production during infection in vivo. FASEB J. 14:1447-1454. https://pubmed.ncbi.nlm.nih.gov/10877838
- Akaike TY, Noguchi, S Ijiri, et al. (1996) Pathogenesis of influenza virus-induced pneumonia: involvement of both nitric oxide and oxygen radicals. Proc. Natl. Acad. Sci. USA. 93:2448-2453. https://pubmed.ncbi.nlm.nih.gov/8637894
- Keshan Disease Research Group. (1979) Observations on effect of sodium selenite in prevention of Keshan Disease. Chin. Med. J. 92:471-476. https://pubmed.ncbi.nlm.nih.gov/114371 http://rs.yiigle.com/CN11215419799207/1069168.htm
- Taylor, E.W. (2009) The oxidative stress-induced niacin sink (OSINS) model for HIV pathogenesis. Toxicology 278:124-130. https://pubmed.ncbi.nlm.nih.gov/19857540
- Wang Y, Huang J, Sun Y, et al. (2021) SARS-CoV-2 suppresses mRNA expression of selenoproteins associated with ferroptosis, endoplasmic reticulum stress and DNA synthesis. Food and Chemical Toxicology 153:112286. https://pubmed.ncbi.nlm.nih.gov/34023458
- Taylor EW, Ruzicka JA, Premadasa L, Zhao L (2016) Cellular Selenoprotein mRNA Tethering via Antisense Interactions with Ebola and HIV-1 mRNAs May Impact Host Selenium Biochemistry. Cur Top Med Chem. 16:1530-1535. https://pubmed.ncbi.nlm.nih.gov/26369818
- Taylor EW, Radding W. (2020) Understanding Selenium and Glutathione as Antiviral Factors in COVID-19: Does the Viral Mpro Protease Target Host Selenoproteins and Glutathione Synthesis? Front Nutr. 7:143. eCollection 2020. https://pubmed.ncbi.nlm.nih.gov/32984400
- Beck MA, Esworthy RS, Ho YS, Chu FF (1998) Glutathione peroxidase protects mice from viral-induced myocarditis. FASEB J. 12:1143-1149. https://pubmed.ncbi.nlm.nih.gov/9737717 https://www.researchgate.net/publication/13547567_Glutathione_peroxidase_protects_mice_from_viral-induced_myocarditis
- Van der Vliet A, Cross CE. (2000) Oxidants, nitrosants, and the lung. Am. J. Med. 109:398-421. https://pubmed.ncbi.nlm.nih.gov/11020397
- Maeda H, Akaike T. (1991) Oxygen free radicals as pathogenic molecules in viral diseases. Proc. Soc. Exp. Biol. Med. 198:721-727. https://pubmed.ncbi.nlm.nih.gov/1656471
- Beck MA, Handy J, Levander OA. (2000) The role of oxidative stress in viral infections. Ann. N.Y. Acad. Sci. 917:906-912. https://pubmed.ncbi.nlm.nih.gov/11268420
- Grimble RF. (2001) Nutritional modulation of immune function. Proc. Nutr. Soc. 60:389-397. https://pubmed.ncbi.nlm.nih.gov/11681814
- Bhaskaram, P. (2002) Micronutrient malnutrition, infection, and immunity: an overview. Nutr. Rev. 60:S40-S45.
- Field CJ, Johnson IR, Schley PD. (2002) Nutrients and their role in host resistance to infection. J. Leukoc. Biol. 71:16-32. https://pubmed.ncbi.nlm.nih.gov/11781377
- von Essen MR, Kongsbak M, Schjerling P, et al. (2010) Vitamin D controls T cell antigen receptor signaling and activation of human T cells. Nat Immunol. 11:344-349. https://pubmed.ncbi.nlm.nih.gov/20208539
- Arthur JR, McKenzie RC, Beckett GJ. (2003) Selenium in the immune system. J. Nutr. 133:1457S-1459S. https://pubmed.ncbi.nlm.nih.gov/12730442
- Nencioni L, Iuvara A, Aquilano K, et al. (2003) Influenza A virus replication is dependent on an antioxidant pathway that involves GSH and Bcl-2. FASEB J. 17:758-760. https://pubmed.ncbi.nlm.nih.gov/12594179 https://faseb.onlinelibrary.wiley.com/doi/epdf/10.1096/fj.02-0508fje
- Doctor Y, Saul AW, Smith RG (2021) Nutrition to Prevent and Treat COVID-19. Orthomolecular Medicine News Service. http://www.orthomolecular.org/resources/omns/v17n03.shtml (Suggest adding niacin/niacinamide, K2 along with D3, and increased selenium to 200 ug/day to the supplement recommendations listed)
- Passwater M (2020) Do the Math: MATH+ Saves Lives. Orthomolecular Medicine News Service. http://www.orthomolecular.org/resources/omns/v16n55.shtml
- Passwater M (2021) The VICTAS Trial: Designed to Fail.Orthomolecular Medicine News Service. http://www.orthomolecular.org/resources/omns/v17n08.shtml
- Riordan Clinic (2021) High dose vitamin C (IVC) https://riordanclinic.org/what-we-do/high-dose-iv-vitamin-c
- Holford P (2020) Vitamin C for the Prevention and Treatment of Coronavirus. Orthomolecular Medicine News Service. http://orthomolecular.org/resources/omns/v16n36.shtml
- Gonzalez MJ (2020) Personalize Your COVID-19 Prevention: An Orthomolecular Protocol. Orthomolecular Medicine News Service. http://orthomolecular.org/resources/omns/v16n31.shtml
- Rasmussen MPF (2020) Vitamin C Evidence for Treating Complications of COVID-19 and other Viral Infections. Orthomolecular Medicine News Service. http://orthomolecular.org/resources/omns/v16n25.shtml
Nutritional Medicine is Orthomolecular Medicine
Orthomolecular medicine uses safe, effective nutritional therapy to fight illness. For more information: http://www.orthomolecular.org
Orthomolecular Medicine News Service (OMNS)
OMNS free subscription link http://orthomolecular.org/subscribe.html
OMNS archive link http://orthomolecular.org/resources/omns/index.shtml
Find a Doctor
To locate an orthomolecular physician near you: http://orthomolecular.org/resources/omns/v06n09.shtml
The peer-reviewed Orthomolecular Medicine News Service is a non-profit and non-commercial informational resource.
Editorial Review Board
Albert G. B. Amoa, MB.Ch.B, Ph.D. (Ghana)
Seth Ayettey, M.B., Ch.B., Ph.D. (Ghana)
Ilyès Baghli, M.D. (Algeria)
Ian Brighthope, MBBS, FACNEM (Australia)
Gilbert Henri Crussol, D.M.D. (Spain)
Carolyn Dean, M.D., N.D. (USA)
Ian Dettman, Ph.D. (Australia)
Damien Downing, M.B.B.S., M.R.S.B. (United Kingdom)
Susan R. Downs, M.D., M.P.H. (USA)
Ron Ehrlich, B.D.S. (Australia)
Hugo Galindo, M.D. (Colombia)
Martin P. Gallagher, M.D., D.C. (USA)
Michael J. Gonzalez, N.M.D., D.Sc., Ph.D. (Puerto Rico)
William B. Grant, Ph.D. (USA)
Claus Hancke, MD, FACAM (Denmark)
Tonya S. Heyman, M.D. (USA)
Suzanne Humphries, M.D. (USA)
Ron Hunninghake, M.D. (USA)
Bo H. Jonsson, M.D., Ph.D. (Sweden)
Felix I. D. Konotey-Ahulu, MD, FRCP, DTMH (Ghana)
Jeffrey J. Kotulski, D.O. (USA)
Peter H. Lauda, M.D. (Austria)
Alan Lien, Ph.D. (Taiwan)
Homer Lim, M.D. (Philippines)
Stuart Lindsey, Pharm.D. (USA)
Victor A. Marcial-Vega, M.D. (Puerto Rico)
Charles C. Mary, Jr., M.D. (USA)
Mignonne Mary, M.D. (USA)
Jun Matsuyama, M.D., Ph.D. (Japan)
Joseph Mercola, D.O. (USA)
Jorge R. Miranda-Massari, Pharm.D. (Puerto Rico)
Karin Munsterhjelm-Ahumada, M.D. (Finland)
Tahar Naili, M.D. (Algeria)
W. Todd Penberthy, Ph.D. (USA)
Zhiyong Peng, M.D. (China)
Isabella Akyinbah Quakyi, Ph.D. (Ghana)
Selvam Rengasamy, MBBS, FRCOG (Malaysia)
Jeffrey A. Ruterbusch, D.O. (USA)
Gert E. Schuitemaker, Ph.D. (Netherlands)
T.E. Gabriel Stewart, M.B.B.CH. (Ireland)
Thomas L. Taxman, M.D. (USA)
Jagan Nathan Vamanan, M.D. (India)
Garry Vickar, M.D. (USA)
Ken Walker, M.D. (Canada)
Raymond Yuen, MBBS, MMed (Singapore)
Anne Zauderer, D.C. (USA)
Andrew W. Saul, Ph.D. (USA), Editor-In-Chief
Associate Editor: Robert G. Smith, Ph.D. (USA)
Editor, Japanese Edition: Atsuo Yanagisawa, M.D., Ph.D. (Japan)
Editor, Chinese Edition: Richard Cheng, M.D., Ph.D. (USA)
Editor, French Edition: Vladimir Arianoff, M.D. (Belgium)
Editor, Norwegian Edition: Dag Viljen Poleszynski, Ph.D. (Norway)
Editor, Arabic Edition: Moustafa Kamel, R.Ph, P.G.C.M (Egypt)
Editor, Korean Edition: Hyoungjoo Shin, M.D. (South Korea)
Editor, Spanish Edition: Sonia Rita Rial, PhD (Argentina)
Contributing Editor: Thomas E. Levy, M.D., J.D. (USA)
Assistant Editor: Helen Saul Case, M.S. (USA)
Technology Editor: Michael S. Stewart, B.Sc.C.S. (USA)
Associate Technology Editor: Robert C. Kennedy, M.S. (USA)
Legal Consultant: Jason M. Saul, JD (USA)
Comments and media contact:
drsaul@doctoryourself.com OMNS welcomes but is unable to respond to individual reader emails. Reader comments become the property of OMNS and may or may not be used for publication.
Click here to see a web copy of this news release: https://orthomolecular.acemlna.com/p_v.php?l=1&c=200&m=204&s=c7ae1002d2f579a22c16a1b89c854212
Acknowledgement Citation
Republished from orthomolecular.activehosted.com
Research Reveals that Some Individuals have Stronger Natural Defences Against SARS-COV-2
Scientists discover why some individuals have stronger natural defences against SARS-COV-2 A new study has revealed key insights into the natural human antiviral defences against SARS-CoV-2, the virus that causes COVID-19. The research, published in the journal Science and led by a team of scientists at the MRC-University of Glasgow Centre for Virus Research, sheds new light on why some people are naturally more resistant to serious SARS-CoV-2 infection – and how, in the future, the coronavirus might overcome this resistance. A link to the study can be found here: https://www.science.org/doi/10.1126/science.abj3624 [1]
COVID-19 is spread from person to person after the virus, shed by an infected person, infects the cells of a new host. Once infected, our cells try to fight off the invading virus and scientists already know that this works better is some people, making their experience of the disease less severe. However, until now, that anti-viral response – and its effect on the virus SARS-CoV-2 – hasn’t been well-understood.
In the study, the scientists reveal that some people have a version of a gene, called OAS1, that potently inhibits SARS-CoV-2. The study showed that, while some people can express a more protective ‘prenylated’ version of the OAS1 gene, other people express a version of this gene which does not detect SARS-CoV-2. Inside cells, coronaviruses hide and replicate inside vesicles coated with lipid (fat). Prenylation is the addition of a single molecule of lipid (fat) to a protein – and it’s this technical difference that allows prenylated OAS1 to ‘seek out’ the invading virus and ‘sound the alarm’.
The study found that, in hospitalized patients, expression of a prenylated version of this gene was associated with protection from severe COVID-19, which suggests this antiviral defence is a major component of a protective antiviral response; and is likely to have offered protection to many people during the course of the pandemic. The study also found that hospitalized COVID-19 patients with the ‘bad’ form of OAS1 had worse clinical outcomes compared to those who expressed the protective prenylated version of OAS1. Severe disease was significantly more frequent with ICU admission or death being approximately 1.6 times more likely in these patients.
Interestingly, the researchers also found that, approximately 55 million years ago, there was a removal of this protective gene in horseshoe bats – the presumed source of SARS-CoV-2) – so therefore SARS-CoV-2 never had to adapt to evade this defence. As the protective prenylated OAS1 gene is widespread in animals, the billions of people that lack this protective gene could make humans particularly vulnerable to the spill over of coronaviruses from horseshoe bats.
Sam Wilson said:
“We know viruses adapt, and even SARS-CoV-2 has likely adapted to replicate in the animal reservoir(s) in which it circulates. Cross-species transmission to humans exposed the virus SARS-CoV-2 to a new repertoire of antiviral defences, some of which SARS-CoV-2 may not know how to evade.
“What our study shows us is that the coronavirus that caused the SARS outbreak in 2003 learned to evade prenylated OAS1. If SARS-CoV-2 variants learn the same trick, they could be substantially more pathogenic and transmissible in unvaccinated populations. This reinforces the need to continually monitor the emergence of new SARS-CoV-2 variants.”
The study, ‘A Prenylated dsRNA Sensor Protects Against Severe COVID-19,’ is published in Science.[1] The study was predominantly funded by the Medical Research Council, Wellcome, and UKRI.
Reference
Wickenhagen A, Sugrue E, Lytras S, Kuchi S, Noerenbert M, Turnbull M, Loney C, Herder V, Allan J et al. A prenylated dsRNA sensor protects against severe COVID-19. Science. DOI: 10.1126/science.abj3624. https://www.science.org/doi/10.1126/science.abj3624 . 28 Sep 2021.
Further Information
For more information contact Elizabeth McMeekin or Ali Howard in the University of Glasgow Communications and Public Affairs Office on Tel: 0141 330 4831 or Tel: 0141 330 6557; or email Elizabeth.mcmeekin@glasgow.ac.uk or ali.howard@glasgow.ac.uk
Comments:
-
No Article Comments available